Unit 23: Organic Chemistry (NOT COVERED)
Hydrocarbons, learning objectives.
By the end of this section, you will be able to:
- Explain the importance of hydrocarbons and the reason for their diversity
- Name saturated and unsaturated hydrocarbons, and molecules derived from them
- Describe the reactions characteristic of saturated and unsaturated hydrocarbons
- Identify structural and geometric isomers of hydrocarbons
The largest database [1] of organic compounds lists about 10 million substances, which include compounds originating from living organisms and those synthesized by chemists. The number of potential organic compounds has been estimated [2] at 10 60 —an astronomically high number. The existence of so many organic molecules is a consequence of the ability of carbon atoms to form up to four strong bonds to other carbon atoms, resulting in chains and rings of many different sizes, shapes, and complexities.
The simplest organic compounds contain only the elements carbon and hydrogen, and are called hydrocarbons. Even though they are composed of only two types of atoms, there is a wide variety of hydrocarbons because they may consist of varying lengths of chains, branched chains, and rings of carbon atoms, or combinations of these structures. In addition, hydrocarbons may differ in the types of carbon-carbon bonds present in their molecules. Many hydrocarbons are found in plants, animals, and their fossils; other hydrocarbons have been prepared in the laboratory. We use hydrocarbons every day, mainly as fuels, such as natural gas, acetylene, propane, butane, and the principal components of gasoline, diesel fuel, and heating oil. The familiar plastics polyethylene, polypropylene, and polystyrene are also hydrocarbons. We can distinguish several types of hydrocarbons by differences in the bonding between carbon atoms. This leads to differences in geometries and in the hybridization of the carbon orbitals.
Alkanes , or saturated hydrocarbons , contain only single covalent bonds between carbon atoms. Each of the carbon atoms in an alkane has sp 3 hybrid orbitals and is bonded to four other atoms, each of which is either carbon or hydrogen. The Lewis structures and models of methane, ethane, and pentane are illustrated in Figure 1. Carbon chains are usually drawn as straight lines in Lewis structures, but one has to remember that Lewis structures are not intended to indicate the geometry of molecules. Notice that the carbon atoms in the structural models (the ball-and-stick and space-filling models) of the pentane molecule do not lie in a straight line. Because of the sp 3 hybridization, the bond angles in carbon chains are close to 109.5°, giving such chains in an alkane a zigzag shape.
The structures of alkanes and other organic molecules may also be represented in a less detailed manner by condensed structural formulas (or simply, condensed formulas ). Instead of the usual format for chemical formulas in which each element symbol appears just once, a condensed formula is written to suggest the bonding in the molecule. These formulas have the appearance of a Lewis structure from which most or all of the bond symbols have been removed. Condensed structural formulas for ethane and pentane are shown at the bottom of Figure 1, and several additional examples are provided in the exercises at the end of this chapter.

Figure 1. Pictured are the Lewis structures, ball-and-stick models, and space-filling models for molecules of methane, ethane, and pentane.
A common method used by organic chemists to simplify the drawings of larger molecules is to use a skeletal structure (also called a line-angle structure). In this type of structure, carbon atoms are not symbolized with a C, but represented by each end of a line or bend in a line. Hydrogen atoms are not drawn if they are attached to a carbon. Other atoms besides carbon and hydrogen are represented by their elemental symbols. Figure 2 shows three different ways to draw the same structure.

Figure 2. The same structure can be represented three different ways: an expanded formula, a condensed formula, and a skeletal structure.

Example 1: Drawing Skeletal Structures
Draw the skeletal structures for these two molecules:

Each carbon atom is converted into the end of a line or the place where lines intersect. All hydrogen atoms attached to the carbon atoms are left out of the structure (although we still need to recognize they are there):

Check Your Learning

Example 2: Interpreting Skeletal Structures
Identify the chemical formula of the molecule represented here:

There are eight places where lines intersect or end, meaning that there are eight carbon atoms in the molecule. Since we know that carbon atoms tend to make four bonds, each carbon atom will have the number of hydrogen atoms that are required for four bonds. This compound contains 16 hydrogen atoms for a molecular formula of C 8 H 16 .
Location of the hydrogen atoms:

All alkanes are composed of carbon and hydrogen atoms, and have similar bonds, structures, and formulas; noncyclic alkanes all have a formula of C n H 2n+2 . The number of carbon atoms present in an alkane has no limit. Greater numbers of atoms in the molecules will lead to stronger intermolecular attractions (dispersion forces) and correspondingly different physical properties of the molecules. Properties such as melting point and boiling point (Table 1) usually change smoothly and predictably as the number of carbon and hydrogen atoms in the molecules change.
Hydrocarbons with the same formula, including alkanes, can have different structures. For example, two alkanes have the formula C 4 H 10 : They are called n -butane and 2-methylpropane (or isobutane), and have the following Lewis structures:

The compounds n -butane and 2-methylpropane are structural isomers (the term constitutional isomers is also commonly used). Constitutional isomers have the same molecular formula but different spatial arrangements of the atoms in their molecules. The n -butane molecule contains an unbranched chain , meaning that no carbon atom is bonded to more than two other carbon atoms. We use the term normal , or the prefix n , to refer to a chain of carbon atoms without branching. The compound 2–methylpropane has a branched chain (the carbon atom in the center of the Lewis structure is bonded to three other carbon atoms)
Identifying isomers from Lewis structures is not as easy as it looks. Lewis structures that look different may actually represent the same isomers. For example, the three structures in Figure 3 all represent the same molecule, n -butane, and hence are not different isomers. They are identical because each contains an unbranched chain of four carbon atoms.

Figure 3. These three representations of the structure of n-butane are not isomers because they all contain the same arrangement of atoms and bonds.
The Basics of Organic Nomenclature: Naming Alkanes
The International Union of Pure and Applied Chemistry ( IUPAC ) has devised a system of nomenclature that begins with the names of the alkanes and can be adjusted from there to account for more complicated structures. The nomenclature for alkanes is based on two rules:
- To name an alkane, first identify the longest chain of carbon atoms in its structure. A two-carbon chain is called ethane; a three-carbon chain, propane; and a four-carbon chain, butane. Longer chains are named as follows: pentane (five-carbon chain), hexane (6), heptane (7), octane (8), nonane (9), and decane (10). These prefixes can be seen in the names of the alkanes described in Table 1.
- Add prefixes to the name of the longest chain to indicate the positions and names of substituents . Substituents are branches or functional groups that replace hydrogen atoms on a chain. The position of a substituent or branch is identified by the number of the carbon atom it is bonded to in the chain. We number the carbon atoms in the chain by counting from the end of the chain nearest the substituents. Multiple substituents are named individually and placed in alphabetical order at the front of the name.

When more than one substituent is present, either on the same carbon atom or on different carbon atoms, the substituents are listed alphabetically. Because the carbon atom numbering begins at the end closest to a substituent, the longest chain of carbon atoms is numbered in such a way as to produce the lowest number for the substituents. The ending – o replaces – ide at the end of the name of an electronegative substituent (in ionic compounds, the negatively charged ion ends with -ide like chloride; in organic compounds, such atoms are treated as substituents and the -o ending is used). The number of substituents of the same type is indicated by the prefixes di- (two), tri- (three), tetra- (four), etc. (for example, difluoro- indicates two fluoride substituents).
Example 3: Naming Halogen-substituted Alkanes
Name the molecule whose structure is shown here:

The four-carbon chain is numbered from the end with the chlorine atom. This puts the substituents on positions 1 and 2 (numbering from the other end would put the substituents on positions 3 and 4). Four carbon atoms means that the base name of this compound will be butane. The bromine at position 2 will be described by adding 2-bromo-; this will come at the beginning of the name, since bromo- comes before chloro- alphabetically. The chlorine at position 1 will be described by adding 1-chloro-, resulting in the name of the molecule being 2-bromo-1-chlorobutane.
Name the following molecule:

We call a substituent that contains one less hydrogen than the corresponding alkane an alkyl group. The name of an alkyl group is obtained by dropping the suffix -ane of the alkane name and adding – yl :

The open bonds in the methyl and ethyl groups indicate that these alkyl groups are bonded to another atom.
Example 4: Naming Substituted Alkanes

The longest carbon chain runs horizontally across the page and contains six carbon atoms (this makes the base of the name hexane, but we will also need to incorporate the name of the branch). In this case, we want to number from right to left (as shown by the red numbers) so the branch is connected to carbon 3 (imagine the numbers from left to right—this would put the branch on carbon 4, violating our rules). The branch attached to position 3 of our chain contains two carbon atoms (numbered in blue)—so we take our name for two carbons eth- and attach -yl at the end to signify we are describing a branch. Putting all the pieces together, this molecule is 3-ethylhexane.

Some hydrocarbons can form more than one type of alkyl group when the hydrogen atoms that would be removed have different “environments” in the molecule. This diversity of possible alkyl groups can be identified in the following way: The four hydrogen atoms in a methane molecule are equivalent; they all have the same environment. They are equivalent because each is bonded to a carbon atom (the same carbon atom) that is bonded to three hydrogen atoms. (It may be easier to see the equivalency in the ball and stick models in Figure 1. Removal of any one of the four hydrogen atoms from methane forms a methyl group. Likewise, the six hydrogen atoms in ethane are equivalent (Figure 1) and removing any one of these hydrogen atoms produces an ethyl group. Each of the six hydrogen atoms is bonded to a carbon atom that is bonded to two other hydrogen atoms and a carbon atom. However, in both propane and 2–methylpropane, there are hydrogen atoms in two different environments, distinguished by the adjacent atoms or groups of atoms:

Each of the six equivalent hydrogen atoms of the first type in propane and each of the nine equivalent hydrogen atoms of that type in 2-methylpropane (all shown in black) are bonded to a carbon atom that is bonded to only one other carbon atom. The two purple hydrogen atoms in propane are of a second type. They differ from the six hydrogen atoms of the first type in that they are bonded to a carbon atom bonded to two other carbon atoms. The green hydrogen atom in 2-methylpropane differs from the other nine hydrogen atoms in that molecule and from the purple hydrogen atoms in propane. The green hydrogen atom in 2-methylpropane is bonded to a carbon atom bonded to three other carbon atoms. Two different alkyl groups can be formed from each of these molecules, depending on which hydrogen atom is removed. The names and structures of these and several other alkyl groups are listed in Figure 4.

Figure 4. This listing gives the names and formulas for various alkyl groups formed by the removal of hydrogen atoms from different locations.
Note that alkyl groups do not exist as stable independent entities. They are always a part of some larger molecule. The location of an alkyl group on a hydrocarbon chain is indicated in the same way as any other substituent:

Alkanes are relatively stable molecules, but heat or light will activate reactions that involve the breaking of C–H or C–C single bonds. Combustion is one such reaction:
[latex]{\text{CH}}_{4}\left(g\right)+2{\text{O}}_{2}\left(g\right)\rightarrow{\text{CO}}_{2}\left(g\right)+2{\text{H}}_{2}\text{O}\left(\text{g}\right)[/latex]
Alkanes burn in the presence of oxygen, a highly exothermic oxidation-reduction reaction that produces carbon dioxide and water. As a consequence, alkanes are excellent fuels. For example, methane, CH 4 , is the principal component of natural gas. Butane, C 4 H 10 , used in camping stoves and lighters is an alkane. Gasoline is a liquid mixture of continuous- and branched-chain alkanes, each containing from five to nine carbon atoms, plus various additives to improve its performance as a fuel. Kerosene, diesel oil, and fuel oil are primarily mixtures of alkanes with higher molecular masses. The main source of these liquid alkane fuels is crude oil, a complex mixture that is separated by fractional distillation. Fractional distillation takes advantage of differences in the boiling points of the components of the mixture (see Figure 5). You may recall that boiling point is a function of intermolecular interactions, which was discussed in the chapter on solutions and colloids.

Figure 5. In a column for the fractional distillation of crude oil, oil heated to about 425 °C in the furnace vaporizes when it enters the base of the tower. The vapors rise through bubble caps in a series of trays in the tower. As the vapors gradually cool, fractions of higher, then of lower, boiling points condense to liquids and are drawn off. (credit left: modification of work by Luigi Chiesa)
In a substitution reaction , another typical reaction of alkanes, one or more of the alkane’s hydrogen atoms is replaced with a different atom or group of atoms. No carbon-carbon bonds are broken in these reactions, and the hybridization of the carbon atoms does not change. For example, the reaction between ethane and molecular chlorine depicted here is a substitution reaction:

The C–Cl portion of the chloroethane molecule is an example of a functional group , the part or moiety of a molecule that imparts a specific chemical reactivity. The types of functional groups present in an organic molecule are major determinants of its chemical properties and are used as a means of classifying organic compounds as detailed in the remaining sections of this chapter.
Want more practice naming alkanes? Watch this brief video tutorial to review the nomenclature process.
Organic compounds that contain one or more double or triple bonds between carbon atoms are described as unsaturated. You have likely heard of unsaturated fats. These are complex organic molecules with long chains of carbon atoms, which contain at least one double bond between carbon atoms. Unsaturated hydrocarbon molecules that contain one or more double bonds are called alkenes . Carbon atoms linked by a double bond are bound together by two bonds, one σ bond and one π bond. Double and triple bonds give rise to a different geometry around the carbon atom that participates in them, leading to important differences in molecular shape and properties. The differing geometries are responsible for the different properties of unsaturated versus saturated fats.
Ethene, C 2 H 4 , is the simplest alkene. Each carbon atom in ethene, commonly called ethylene, has a trigonal planar structure. The second member of the series is propene (propylene) (Figure 6); the butene isomers follow in the series. Four carbon atoms in the chain of butene allows for the formation of isomers based on the position of the double bond, as well as a new form of isomerism.

Figure 6. Expanded structures, ball-and-stick structures, and space-filling models for the alkenes ethene, propene, and 1-butene are shown.
Ethylene (the common industrial name for ethene) is a basic raw material in the production of polyethylene and other important compounds. Over 135 million tons of ethylene were produced worldwide in 2010 for use in the polymer, petrochemical, and plastic industries. Ethylene is produced industrially in a process called cracking, in which the long hydrocarbon chains in a petroleum mixture are broken into smaller molecules.
Everyday Life: Recycling Plastics
Polymers (from Greek words poly meaning “many” and mer meaning “parts”) are large molecules made up of repeating units, referred to as monomers. Polymers can be natural (starch is a polymer of sugar residues and proteins are polymers of amino acids) or synthetic [like polyethylene, polyvinyl chloride (PVC), and polystyrene]. The variety of structures of polymers translates into a broad range of properties and uses that make them integral parts of our everyday lives. Adding functional groups to the structure of a polymer can result in significantly different properties (see Chemistry in Everyday Life: Kevlar, later in this chapter).
An example of a polymerization reaction is shown in Figure 7. The monomer ethylene (C 2 H 4 ) is a gas at room temperature, but when polymerized, using a transition metal catalyst, it is transformed into a solid material made up of long chains of –CH 2 – units called polyethylene. Polyethylene is a commodity plastic used primarily for packaging (bags and films).

Figure 7. The reaction for the polymerization of ethylene to polyethylene is shown.
Polyethylene is a member of one subset of synthetic polymers classified as plastics. Plastics are synthetic organic solids that can be molded; they are typically organic polymers with high molecular masses. Most of the monomers that go into common plastics (ethylene, propylene, vinyl chloride, styrene, and ethylene terephthalate) are derived from petrochemicals and are not very biodegradable, making them candidate materials for recycling. Recycling plastics helps minimize the need for using more of the petrochemical supplies and also minimizes the environmental damage caused by throwing away these nonbiodegradable materials.
Plastic recycling is the process of recovering waste, scrap, or used plastics, and reprocessing the material into useful products. For example, polyethylene terephthalate (soft drink bottles) can be melted down and used for plastic furniture, in carpets, or for other applications. Other plastics, like polyethylene (bags) and polypropylene (cups, plastic food containers), can be recycled or reprocessed to be used again. Many areas of the country have recycling programs that focus on one or more of the commodity plastics that have been assigned a recycling code (see Figure 8). These operations have been in effect since the 1970s and have made the production of some plastics among the most efficient industrial operations today.

Figure 8. Each type of recyclable plastic is imprinted with a code for easy identification.
The name of an alkene is derived from the name of the alkane with the same number of carbon atoms. The presence of the double bond is signified by replacing the suffix -ane with the suffix -ene . The location of the double bond is identified by naming the smaller of the numbers of the carbon atoms participating in the double bond:

Isomers of Alkenes
Molecules of 1-butene and 2-butene are structural isomers; the arrangement of the atoms in these two molecules differs. As an example of arrangement differences, the first carbon atom in 1-butene is bonded to two hydrogen atoms; the first carbon atom in 2-butene is bonded to three hydrogen atoms.
The compound 2-butene and some other alkenes also form a second type of isomer called a geometric isomer. In a set of geometric isomers, the same types of atoms are attached to each other in the same order, but the geometries of the two molecules differ. Geometric isomers of alkenes differ in the orientation of the groups on either side of a [latex]\text{C}=\text{C}[/latex] bond.
Carbon atoms are free to rotate around a single bond but not around a double bond; a double bond is rigid. This makes it possible to have two isomers of 2-butene, one with both methyl groups on the same side of the double bond and one with the methyl groups on opposite sides. When structures of butene are drawn with 120° bond angles around the sp 2 -hybridized carbon atoms participating in the double bond, the isomers are apparent. The 2-butene isomer in which the two methyl groups are on the same side is called a cis -isomer; the one in which the two methyl groups are on opposite sides is called a trans -isomer (Figure 9). The different geometries produce different physical properties, such as boiling point, that may make separation of the isomers possible:

Figure 9. These molecular models show the structural and geometric isomers of butene.
Alkenes are much more reactive than alkanes because the [latex]\text{C}=\text{C}[/latex] moiety is a reactive functional group. A π bond, being a weaker bond, is disrupted much more easily than a σ bond. Thus, alkenes undergo a characteristic reaction in which the π bond is broken and replaced by two σ bonds. This reaction is called an addition reaction. The hybridization of the carbon atoms in the double bond in an alkene changes from sp 2 to sp 3 during an addition reaction. For example, halogens add to the double bond in an alkene instead of replacing hydrogen, as occurs in an alkane:

Example 5: Alkene Reactivity and Naming
Provide the IUPAC names for the reactant and product of the halogenation reaction shown here:

The reactant is a five-carbon chain that contains a carbon-carbon double bond, so the base name will be pentene. We begin counting at the end of the chain closest to the double bond—in this case, from the left—the double bond spans carbons 2 and 3, so the name becomes 2-pentene. Since there are two carbon-containing groups attached to the two carbon atoms in the double bond—and they are on the same side of the double bond—this molecule is the cis- isomer, making the name of the starting alkene cis -2-pentene. The product of the halogenation reaction will have two chlorine atoms attached to the carbon atoms that were a part of the carbon-carbon double bond:

This molecule is now a substituted alkane and will be named as such. The base of the name will be pentane. We will count from the end that numbers the carbon atoms where the chlorine atoms are attached as 2 and 3, making the name of the product 2,3-dichloropentane.
Provide names for the reactant and product of the reaction shown:

Hydrocarbon molecules with one or more triple bonds are called alkynes ; they make up another series of unsaturated hydrocarbons. Two carbon atoms joined by a triple bond are bound together by one σ bond and two π bonds. The sp -hybridized carbons involved in the triple bond have bond angles of 180°, giving these types of bonds a linear, rod-like shape.
The simplest member of the alkyne series is ethyne, C 2 H 2 , commonly called acetylene. The Lewis structure for ethyne, a linear molecule, is:

The IUPAC nomenclature for alkynes is similar to that for alkenes except that the suffix -yne is used to indicate a triple bond in the chain. For example, [latex]{\text{CH}}_{3}{\text{CH}}_{2}\text{C}\equiv \text{CH}[/latex] is called 1-butyne.
Example 6: Structure of Alkynes
Describe the geometry and hybridization of the carbon atoms in the following molecule:

Identify the hybridization and bond angles at the carbon atoms in the molecule shown:

Chemically, the alkynes are similar to the alkenes. Since the [latex]\text{C}\equiv \text{C}[/latex] functional group has two π bonds, alkynes typically react even more readily, and react with twice as much reagent in addition reactions. The reaction of acetylene with bromine is a typical example:

Acetylene and the other alkynes also burn readily. An acetylene torch takes advantage of the high heat of combustion for acetylene.
Aromatic Hydrocarbons
Benzene, C 6 H 6 , is the simplest member of a large family of hydrocarbons, called aromatic hydrocarbons . These compounds contain ring structures and exhibit bonding that must be described using the resonance hybrid concept of valence bond theory or the delocalization concept of molecular orbital theory. (To review these concepts, refer to the earlier chapters on chemical bonding). The resonance structures for benzene, C 6 H 6 , are:

Valence bond theory describes the benzene molecule and other planar aromatic hydrocarbon molecules as hexagonal rings of sp 2 -hybridized carbon atoms with the unhybridized p orbital of each carbon atom perpendicular to the plane of the ring. Three valence electrons in the sp 2 hybrid orbitals of each carbon atom and the valence electron of each hydrogen atom form the framework of σ bonds in the benzene molecule. The fourth valence electron of each carbon atom is shared with an adjacent carbon atom in their unhybridized p orbitals to yield the π bonds.

Figure 10. This condensed formula shows the unique bonding structure of benzene.
Benzene does not, however, exhibit the characteristics typical of an alkene. Each of the six bonds between its carbon atoms is equivalent and exhibits properties that are intermediate between those of a C–C single bond and a [latex]\text{C}=\text{C}[/latex] double bond. To represent this unique bonding, structural formulas for benzene and its derivatives are typically drawn with single bonds between the carbon atoms and a circle within the ring as shown in Figure 10.
There are many derivatives of benzene. The hydrogen atoms can be replaced by many different substituents. Aromatic compounds more readily undergo substitution reactions than addition reactions; replacement of one of the hydrogen atoms with another substituent will leave the delocalized double bonds intact. The following are typical examples of substituted benzene derivatives:

Toluene and xylene are important solvents and raw materials in the chemical industry. Styrene is used to produce the polymer polystyrene.
Example 7: Structure of Aromatic Hydrocarbons
One possible isomer created by a substitution reaction that replaces a hydrogen atom attached to the aromatic ring of toluene with a chlorine atom is shown here. Draw two other possible isomers in which the chlorine atom replaces a different hydrogen atom attached to the aromatic ring:

Since the six-carbon ring with alternating double bonds is necessary for the molecule to be classified as aromatic, appropriate isomers can be produced only by changing the positions of the chloro-substituent relative to the methyl-substituent:

Draw three isomers of a six-membered aromatic ring compound substituted with two bromines.

Key Concepts and Summary
Strong, stable bonds between carbon atoms produce complex molecules containing chains, branches, and rings. The chemistry of these compounds is called organic chemistry. Hydrocarbons are organic compounds composed of only carbon and hydrogen. The alkanes are saturated hydrocarbons—that is, hydrocarbons that contain only single bonds. Alkenes contain one or more carbon-carbon double bonds. Alkynes contain one or more carbon-carbon triple bonds. Aromatic hydrocarbons contain ring structures with delocalized π electron systems.
- What is the difference between the hybridization of carbon atoms’ valence orbitals in saturated and unsaturated hydrocarbons?
- On a microscopic level, how does the reaction of bromine with a saturated hydrocarbon differ from its reaction with an unsaturated hydrocarbon? How are they similar?
- On a microscopic level, how does the reaction of bromine with an alkene differ from its reaction with an alkyne? How are they similar?
- Explain why unbranched alkenes can form geometric isomers while unbranched alkanes cannot. Does this explanation involve the macroscopic domain or the microscopic domain?

- How does the carbon-atom hybridization change when polyethylene is prepared from ethylene?
- 3-methylpentane
- cis -3-hexene
- 4-methyl-1-pentene
- 4-methyl-2-pentyne
- 3-methylhexane
- trans -3-heptene
- 4-methyl-1-hexene
- 3,4-dimethyl-1-pentyne
- CH 3 CH 2 CBr 2 CH 3
- (CH 3 ) 3 CCl

- [latex]{\text{CH}}_{3}{\text{CH}}_{2}\text{C}\equiv {\text{CH CH}}_{3}{\text{CH}}_{2}\text{C}\equiv \text{CH}[/latex]

- [latex]{\left({\text{CH}}_{3}\right)}_{2}{\text{CHCH}}_{2}\text{CH}={\text{CH}}_{2}[/latex]
- (CH 3 ) 2 CHF
- CH 3 CHClCHClCH 3

- [latex]{\text{CH}}_{3}{\text{CH}}_{2}\text{CH}={\text{CHCH}}_{3}[/latex]

- [latex]{\left({\text{CH}}_{3}\right)}_{3}{\text{CCH}}_{2}\text{C}\equiv \text{CH}[/latex]
- Butane is used as a fuel in disposable lighters. Write the Lewis structure for each isomer of butane.
- Write Lewis structures and name the five structural isomers of hexane.
- Write Lewis structures for the cis–trans isomers of [latex]{\text{CH}}_{3}\text{CH}=\text{CHCl.}[/latex]
- Write structures for the three isomers of the aromatic hydrocarbon xylene, C 6 H 4 (CH 3 ) 2 .

- What is the IUPAC name for the compound?
- Name the other isomers that contain a five-carbon chain with three methyl substituents.
- Write Lewis structures and IUPAC names for the alkyne isomers of C 4 H 6 .
- Write Lewis structures and IUPAC names for all isomers of C 4 H 9 Cl.
- Name and write the structures of all isomers of the propyl and butyl alkyl groups.
- Write the structures for all the isomers of the –C 5 H 11 alkyl group.
- cis -1-chloro-2-bromoethene
- trans – 6 -ethyl-7-methyl-2-octene
- Benzene is one of the compounds used as an octane enhancer in unleaded gasoline. It is manufactured by the catalytic conversion of acetylene to benzene: [latex]3{\text{C}}_{2}{\text{H}}_{2}\rightarrow{\text{C}}_{6}{\text{H}}_{6}[/latex]. Draw Lewis structures for these compounds, with resonance structures as appropriate, and determine the hybridization of the carbon atoms in each.
- Teflon is prepared by the polymerization of tetrafluoroethylene. Write the equation that describes the polymerization using Lewis symbols.
- 1 mol of 1-butyne reacts with 2 mol of iodine.
- Pentane is burned in air.
- 2-butene reacts with chlorine.
- benzene burns in air.
- What mass of 2-bromopropane could be prepared from 25.5 g of propene? Assume a 100% yield of product.
- What is the empirical formula of the compound of silver and carbon?
- The production of acetylene on addition of HCl to the compound of silver and carbon suggests that the carbon is present as the acetylide ion, [latex]{\text{C}}_{2}{}^{2-}[/latex] . Write the formula of the compound showing the acetylide ion.
- Ethylene can be produced by the pyrolysis of ethane: [latex]{\text{C}}_{2}{\text{H}}_{6}\rightarrow{\text{C}}_{2}{\text{H}}_{4}+{\text{H}}_{2}[/latex]. How many kilograms of ethylene is produced by the pyrolysis of [latex]1.000\times {10}^{3}\text{kg}[/latex] of ethane, assuming a 100.0% yield?
1. There are several sets of answers; one is:

3. Both reactions result in bromine being incorporated into the structure of the product. The difference is the way in which that incorporation takes place. In the saturated hydrocarbon, an existing C–H bond is broken, and a bond between the C and the Br can then be formed. In the unsaturated hydrocarbon, the only bond broken in the hydrocarbon is the π bond whose electrons can be used to form a bond to one of the bromine atoms in Br 2 (the electrons from the Br–Br bond form the other C–Br bond on the other carbon that was part of the π bond in the starting unsaturated hydrocarbon).
5. Unbranched alkanes have free rotation about the C–C bonds, yielding all orientations of the substituents about these bonds equivalent, interchangeable by rotation. In the unbranched alkenes, the inability to rotate about the C=C bond results in fixed (unchanging) substituent orientations, thus permitting different isomers. Since these concepts pertain to phenomena at the molecular level, this explanation involves the microscopic domain.
7. They are the same compound because each is a saturated hydrocarbon containing an unbranched chain of six carbon atoms.
9. The Lewis structures and molecular formulas are as follows:

11. The IUPAC names are as follows:
- 2,2-dibromobutane
- 2-chloro-2-methylpropane
- 2-methylbutane
- 4-fluoro-4-methyl-1-octyne
- trans -1-chloropropene
- 5-methyl-1-pentene

17. The answers are as follows:
- 2,2,4-trimethylpentane

In acetylene, the bonding uses sp hybrids on carbon atoms and s orbitals on hydrogen atoms. In benzene, the carbon atoms are sp 2 hybridized.
25. The balanced equations are as follows:

27. [latex]{\text{C}}_{3}{\text{H}}_{7}\text{Br mass}=22.5\text{g}\times \frac{1\text{mol}{\text{C}}_{3}{\text{H}}_{6}}{48.081\text{g}{\text{C}}_{3}{\text{H}}_{6}}\times \frac{1\text{mol}{\text{C}}_{3}{\text{H}}_{7}\text{Br}}{1\text{mol}{\text{C}}_{3}{\text{H}}_{6}}\times \frac{122.993\text{g}{\text{C}}_{3}{\text{H}}_{7}\text{Br}}{1\text{mol}{\text{C}}_{3}{\text{H}}_{7}\text{Br}}=65.2\text{g}[/latex]
29. [latex]1{\text{C}}_{2}{\text{H}}_{6}\rightarrow 1{\text{C}}_{2}{\text{H}}_{4}[/latex]
[latex]\begin{array}{rcl}\text{mass of ethylene}&=&1\times{10}^{3}\text{kg}\times\frac{1\text{mol}}{30.07\text{g}}\times \frac{28.05\text{g}}{1\text{mol}}\\{}&=& 9.328\times{10}^{2}\text{kg}\end{array}[/latex]
alkane: molecule consisting of only carbon and hydrogen atoms connected by single (σ) bonds
alkene: molecule consisting of carbon and hydrogen containing at least one carbon-carbon double bond
alkyl group: substituent, consisting of an alkane missing one hydrogen atom, attached to a larger structure
alkyne: molecule consisting of carbon and hydrogen containing at least one carbon-carbon triple bond
aromatic hydrocarbon: cyclic molecule consisting of carbon and hydrogen with delocalized alternating carbon-carbon single and double bonds, resulting in enhanced stability
functional group: part of an organic molecule that imparts a specific chemical reactivity to the molecule
organic compound: natural or synthetic compound that contains carbon
saturated hydrocarbon: molecule containing carbon and hydrogen that has only single bonds between carbon atoms
skeletal structure: shorthand method of drawing organic molecules in which carbon atoms are represented by the ends of lines and bends in between lines, and hydrogen atoms attached to the carbon atoms are not shown (but are understood to be present by the context of the structure)
substituent: branch or functional group that replaces hydrogen atoms in a larger hydrocarbon chain
substitution reaction: reaction in which one atom replaces another in a molecule
- This is the Beilstein database, now available through the Reaxys site ( http://www.elsevier.com/online-tools/reaxys ). ↵
- Peplow, Mark. “Organic Synthesis: The Robo-Chemist,” Nature 512 (2014): 20–2. ↵
- Physical properties for C 4 H 10 and heavier molecules are those of the normal isomer, n-butane, n-pentane, etc. ↵
- STP indicates a temperature of 0 °C and a pressure of 1 atm. ↵
- Chemistry. Provided by : OpenStax College. Located at : http://openstaxcollege.org . License : CC BY: Attribution . License Terms : Download for free at https://openstaxcollege.org/textbooks/chemistry/get
- Hydrocarbons
Unsaturated Hydrocarbons
What are unsaturated hydrocarbons.
Unsaturated hydrocarbons are organic compounds that are entirely made up of carbon and hydrogen atoms and consist of a double or a triple bond between two adjacent carbon atoms. For example, CH 3 CH 2 CH=CH 2 (n-Butylene) & CH 3 CH=CH 2 (Propylene) The difference in the chemical formulae of saturated and unsaturated hydrocarbons is illustrated below.

In the IUPAC nomenclature of unsaturated hydrocarbons, the position of the double or triple bond is either described by a number written before the name of the compound (as in 2,4 pentadiene) or by a number written before the suffix, ‘-ene’ or ‘-yne’ (as in pent-2-ene).
The physical properties of saturated hydrocarbons and unsaturated hydrocarbons are quite similar. These types of hydrocarbons (except aromatic hydrocarbons) are quite reactive and tend to undergo addition reactions with elemental halogens, hydrogen halides, alcohols, and many other compounds.
Table of Contents
Types of unsaturated hydrocarbons, uses of unsaturated hydrocarbons.
Unsaturated hydrocarbons , based on the types of bonds they contain, can be classified into alkenes, alkynes, and aromatic hydrocarbons. The different types of organic compounds that can be classified as unsaturated hydrocarbons are briefly discussed below.
- The hydrocarbons that contain at least one double bond between two adjacent carbon atoms are called alkenes or olefins. The simplest alkene is ethylene (or ethene), given by the chemical formula C 2 H 4 .
- Alkenes containing only one double bond and having no functional groups or substituents attached to them can be generalized to the chemical formula C n H 2n .
- Hydrocarbons containing a minimum of one triple bond between two carbon atoms that are positioned adjacent to each other are referred to as alkynes . The alkyne with the simplest structure is acetylene (systematic IUPAC name: ethyne) with the chemical formula C 2 H 2 .
- The alkynes that have only one carbon-carbon triple bond and have no functional or substituent groups attached to them can be generalized to the chemical formula C n H 2n-2
- Cyclic hydrocarbons that contain at least one double or triple bond between two carbon atoms are also considered to be unsaturated hydrocarbons, one such example being cyclopentene (C 5 H 8 ).
- Although Aromatic Hydrocarbons (ring-shaped hydrocarbons containing delocalised pi electrons) can be considered unsaturated hydrocarbons, but they are generally referred to as aromatic compounds because they are relatively stable and do not share similar properties with other such unsaturated compounds.
In order to check whether a given hydrocarbon is unsaturated, bromine water can be added to it. Should the bromine water become decolourised, the hydrocarbon in question is unsaturated. If a white precipitate is formed, the hydrocarbon sample is phenol or aniline. It can be noted that benzene does not decolourise the bromine water.
Some uses of compounds belonging to the unsaturated hydrocarbon category are listed below.
- Many fruits can be artificially ripened with the help of alkenes.
- Mustard gas, a poisonous gas used in chemical warfare, can be created with the help of alkenes.
- Unsaturated hydrocarbons are extremely useful organic compounds in the manufacturing of plastics.
- LDPE, which is a low-density variation of polyethylene, is used in the manufacturing of grocery bags
- Polystyrene is used in making egg cartons, disposable cups, and other convenient products.
- Industrial chemicals such as alcohol include the usage of alkenes in their manufacturing process.
- Some unsaturated hydrocarbons are used as general anaesthetics.
- Many organic compounds of high industrial importance are manufactured with the help of alkynes.
Recommended Videos
Saturated and unsaturated compounds.

Thus, the general properties, types, and uses of unsaturated hydrocarbons are briefly discussed in this article. For more information on unsaturated hydrocarbons and other types of hydrocarbons, such as aromatic hydrocarbons , register with BYJU’S and download the mobile application on your smartphone.

Put your understanding of this concept to test by answering a few MCQs. Click ‘Start Quiz’ to begin!
Select the correct answer and click on the “Finish” button Check your score and answers at the end of the quiz
Visit BYJU’S for all Chemistry related queries and study materials
Your result is as below
Request OTP on Voice Call
Leave a Comment Cancel reply
Your Mobile number and Email id will not be published. Required fields are marked *
Post My Comment
Its very very simplest way of learning i like it more than more.
What a good ideas. I Understand very well.
- Share Share
Register with BYJU'S & Download Free PDFs
Register with byju's & watch live videos.

Your browser is not supported
Sorry but it looks as if your browser is out of date. To get the best experience using our site we recommend that you upgrade or switch browsers.
Find a solution
- Skip to main content
- Skip to navigation

- Back to parent navigation item
- Primary teacher
- Secondary/FE teacher
- Early career or student teacher
- Higher education
- Curriculum support
- Literacy in science teaching
- Periodic table
- Interactive periodic table
- Climate change and sustainability
- Resources shop
- Collections
- Post-lockdown teaching support
- Remote teaching support
- Starters for ten
- Screen experiments
- Assessment for learning
- Microscale chemistry
- Faces of chemistry
- Classic chemistry experiments
- Nuffield practical collection
- Anecdotes for chemistry teachers
- On this day in chemistry
- Global experiments
- PhET interactive simulations
- Chemistry vignettes
- Context and problem based learning
- Journal of the month
- Chemistry and art
- Art analysis
- Pigments and colours
- Ancient art: today's technology
- Psychology and art theory
- Art and archaeology
- Artists as chemists
- The physics of restoration and conservation
- Ancient Egyptian art
- Ancient Greek art
- Ancient Roman art
- Classic chemistry demonstrations
- In search of solutions
- In search of more solutions
- Creative problem-solving in chemistry
- Solar spark
- Chemistry for non-specialists
- Health and safety in higher education
- Analytical chemistry introductions
- Exhibition chemistry
- Introductory maths for higher education
- Commercial skills for chemists
- Kitchen chemistry
- Journals how to guides
- Chemistry in health
- Chemistry in sport
- Chemistry in your cupboard
- Chocolate chemistry
- Adnoddau addysgu cemeg Cymraeg
- The chemistry of fireworks
- Festive chemistry
- Education in Chemistry
- Teach Chemistry
- On-demand online
- Live online
- Selected PD articles
- PD for primary teachers
- PD for secondary teachers
- What we offer
- Chartered Science Teacher (CSciTeach)
- Teacher mentoring
- UK Chemistry Olympiad
- Who can enter?
- How does it work?
- Resources and past papers
- Top of the Bench
- Schools' Analyst
- Regional support
- Education coordinators
- RSC Yusuf Hamied Inspirational Science Programme
- RSC Education News
- Supporting teacher training
- Interest groups

- More from navigation items
Organic chemistry worksheets | 14–16 years
- 1 Introduction
- 2 Crude oil
- 3 Hydrocarbons
- 4 Cracking hydrocarbons
- 7 Carboxylic acids
- 8 Addition polymerisation
- 9 Condensation polymerisation
- 10 Natural polymers
- 11 Burning hydrocarbons
- 12 Reactions of alkanes and alcohols
Hydrocarbons
By Rob King
- Five out of five
- No comments
Differentiated, editable worksheets providing a wide range of assessment questions exploring hydrocarbons, including structural formulae, writing word equations and balancing symbol equations
In context worksheets
These write-on worksheets will ask learners to use their knowledge of hydrocarbons in an applied context. Calculation questions are included to give opportunities to practise mathematical skills within this topic. Foundation and higher level worksheets are available and fully editable versions give you the flexibility to select the questions most relevant to a particular lesson. The teacher versions (also editable) give answers to all questions.
Foundation level
- Download the student worksheet as MS Word or pdf .
- Download the teacher version including answers to all questions as MS Word or pdf .
Higher level
Knowledge check worksheets.
Provide a series of questions on hydrocarbons to assess learners’ knowledge and understanding of this topic at both foundation and higher levels. The worksheets could be used for individual student work in class or at home. Separate answer sheets allow these resources to be used by teachers or by students during self-assessment of progress.
- Download the student worksheet as MS Word or pdf .
- Download the teacher version including answers to all questions as MS Word or pdf .
- Download the teacher version including answers to all questions as MS Word or pdf .
In context sheet - Hydrocarbons - Foundation - Student
In context sheet - hydrocarbons - foundation - teacher, in context sheet - hydrocarbons - higher - student, in context sheet - hydrocarbons - higher - teacher, knowledge check worksheet - hydrocarbons - foundation - student, knowledge check worksheet - hydrocarbons - foundation - teacher, knowledge check worksheet - hydrocarbons - higher - student, knowledge check worksheet - hydrocarbons - higher - teacher.


Introduction

Cracking hydrocarbons

Carboxylic acids

Addition polymerisation

Condensation polymerisation

Natural polymers

Burning hydrocarbons
Reactions of alkanes and alcohols
- 14-16 years
- Maths skills
- Organic chemistry
Specification
- Standard temperature and pressure (s.t.p). Molar volume ar s.t.p., molar mass, relative molar mass (Mr).
- Alkanes, alkenes and alkynes as homologous series. For alkynes only ethyne to be considered.
- Systematic names, stuctural formulas and structural isomers of alkanes to C-5.
- Chemical reactions can result in a change in temperature. Exothermic and endothermic reactions (and changes of state).
- Combustion of alkanes and other hydrocarbons.
- Recognise and use significant figures as appropriate.
- Use of alkanes as as fuels.
- Awareness of the contributions of chemistry to society, e.g. provision of pure water, fuels, metals, medicines, detergents, enzymes, dyes, paints, semiconductors, liquid crystals and alternative materials, such as plastics, and synthetic fibres; increasi…
- Equations (full and ionic).
- The melting and boiling points of molecular substances are influenced by the strength of these intermolecular forces.
- Alkanes are used as fuels.
- Alkanes are saturated hydrocarbons.
- 6. be able to write balanced full and ionic equations, including state symbols, for chemical reactions
- 8. know the general formula for alkanes
- 9. know that alkanes and cycloalkanes are saturated hydrocarbons
- 1. know that a hydrocarbon is a compound of hydrogen and carbon only
- 20 i. understand, in terms of intermolecular forces, physical properties shown by materials, including: the trends in boiling temperatures of alkanes with increasing chain length
- Chemical reactions can be represented by word equations or equations using symbols and formulae.
- Write formulae and balanced chemical equations for the reactions in this specification.
- Chemical equations can be interpreted in terms of moles.
- Some properties of hydrocarbons depend on the size of their molecules, including boiling point, viscosity and flammability. These properties influence how hydrocarbons are used as fuels.
- Students should be able to recall how boiling point, viscosity and flammability change with increasing molecular size.
- The combustion of hydrocarbon fuels releases energy. During combustion, the carbon and hydrogen in the fuels are oxidised. The complete combustion of a hydrocarbon produces carbon dioxide and water.
- Students should be able to write balanced equations for the complete combustion of hydrocarbons with a given formula.
- Crude oil is a mixture of a very large number of compounds. Most of the compounds in crude oil are hydrocarbons, which are molecules made up of hydrogen and carbon atoms only.
- Most of the hydrocarbons in crude oil are hydrocarbons called alkanes. The general formula for the homologous series of alkanes is C₂H₂ₙ₊₂
- The first four members of the alkanes are methane, ethane, propane and butane.
- Alkane molecules can be represented in the following forms: (C₂H₆ or drawn out version). Students should be able to recognise substances as alkanes given their formulae in these forms.
- Describe the fractions as largely a mixture of compounds of formula CₙH₂ₙ₊₂ which are members of the alkane homologous series.
- Most of the hydrocarbons in crude oil are hydrocarbons called alkanes. The general formula for the homologous series of alkanes is CₙH₂ₙ₊₂
- 0.3 Write balanced chemical equations, including the use of the state symbols (s), (l), (g) and (aq)
- 8.1 Recall that hydrocarbons are compounds that contain carbon and hydrogen only
- 8.4 Recall the names and uses of the following fractions: gases, used in domestic heating and cooking; petrol, used as fuel for cars, kerosene, used as fuel for aircraft; diesel oil, used as fuel for some cars and trains; fuel oil, used as fuel for large…
- 8.5 Explain how hydrocarbons in different fractions differ from each other in: the number of carbon and hydrogen atoms their molecules contain; boiling points; ease of ignition; viscosity; and are mostly members of the alkane homologous series
- 8.6 Explain an homologous series as a series of compounds which: have the same general formula; differ by CH₂ in molecular formulae from neighbouring compounds; show a gradual variation in physical properties, as exemplified by their boiling points; have…
- 8.7 Describe the complete combustion of hydrocarbon fuels as a reaction in which: carbon dioxide and water are produced; energy is given out
- 9.10C Recall the formulae of molecules of the alkanes, methane, ethane, propane and butane, and draw the structures of these molecules, showing all covalent bonds
- 9.11C Explain why the alkanes are saturated hydrocarbons
- 9.16C Describe how the complete combustion of alkanes and alkenes involves the oxidation of the hydrocarbons to produce carbon dioxide and water
- 8.2 Describe crude oil as: a complex mixture of hydrocarbons; containing molecules in which carbon atoms are in chains or rings (names, formulae and structures of specific ring molecules not required); an important source of useful substances…
- C3.4.1 recall that crude oil is a main source of hydrocarbons and is a feedstock for the petrochemical industry
- C3.4.4 describe the fractions of crude oil as largely a mixture of compounds of formula CₙH₂ₙ₊₂ which are members of the alkane homologous series
- C3.4.17 name and draw the structural formulae, using fully displayed formulae, of the first four members of the straight chain alkanes and alkenes, alcohols and carboxylic acids
- C3.4.18 predict the formulae and structures of products of reactions (combustion, addition across a double bond and oxidation of alcohols to carboxylic acids) of the first four and other given members of these homologous series
- C6.1j describe the fractions as largely a mixture of compounds of formula CₙH₂ₙ₊₂ which are members of the alkane homologous series
- C6.1k recall that crude oil is a main source of hydrocarbons and is a feedstock for the petrochemical industry
- C6.2b name and draw the structural formulae, using fully displayed formulae, of the first four members of the straight chain alkanes, alkenes, alcohols and carboxylic acids
- C6.2c predict the formulae and structures of products of reactions of the first four and other given members of the homologous series of alkanes, alkenes and alcohols
- C6.2l describe the fractions as largely a mixture of compounds of formula CₙH₂ₙ₊₂ which are members of the alkane homologous series
- Alkanes: are a homologous series of saturated hydrocarbons
- can be represented by the general formula CₙH₂ₙ₊₂
- Straight-chain and branched alkanes can be systematically named from structural formulae containing no more than 8 carbons in the longest chain.
- Molecular formulae can be written and structural formulae can be drawn, from the systematic names of straight-chain and branched alkanes, containing no more than 8 carbons in the longest chain.
- A homologous series is a family of compounds with the same general formula and similar chemical properties.
- Patterns are often seen in the physical properties of the members of a homologous series.
- The subsequent members of a homologous series show a general increase in their melting and boiling points. This pattern is attributed to increasing strength of the intermolecular forces as the molecular size increases. The type of intermolecular force do…
- Hydrocarbons are compounds containing only hydrogen and carbon atoms.
- Compounds containing only single carbon–carbon bonds are described as saturated.
- Compounds containing at least one carbon–carbon double bond are described as unsaturated.
- The structure of any molecule can be drawn as a full or a shortened structural formula.
- (f) the combustion reactions of hydrocarbons and other fuels
- (k) the general formula CₙH₂ₙ₊₂ for alkanes and CₙH₂ₙ for alkenes
- (l) the names and molecular and structural formulae for simple alkanes and alkenes
- (n) the names of more complex alkanes and alkenes
- 2.5.2 define a homologous series as a family of organic molecules that have the same general formula, show similar chemical properties, show a gradation in their physical properties and differ by a CH₂ group;
- 2.5.3 recall that a hydrocarbon is a compound/molecule consisting of hydrogen and carbon only;
- 2.5.4 recall the general formula of the alkanes and the molecular formula, structural formula and state at room temperature and pressure of methane, ethane, propane and butane;
- 2.5.9 describe the complete combustion of alkanes to produce carbon dioxide and water, including observations and tests to identify the products.
- 1.7.6 calculate the reacting masses of reactants or products, given a balanced symbol equation and using moles and simple ratio, including examples here there is a limiting reactant;
- 1.7.5 calculate the reacting masses of reactants or products, given a balanced symbol equation and using moles and simple ratio, including examples here there is a limiting reactant;
Related articles

Fractional distillation and hydrocarbons | Review my learning worksheets | 14–16 years
By Lyn Nicholls
Identify learning gaps and misconceptions with this set of worksheets offering three levels of support

Composite decking can capture carbon dioxide
2024-04-26T08:58:00Z By Nina Notman
Could our choice of patio decking help reduce the global warming effects of building materials?

Enzymes make dyeing denim more sustainable
2024-04-05T08:06:00Z By Nina Notman
Can researchers find a way to make jeans blue without using toxic chemicals?
No comments yet
Only registered users can comment on this article., more from resources.

Chromatography | Review my learning worksheets | 14–16 years
2024-05-10T13:33:00Z By Lyn Nicholls

Solubility | Review my learning worksheets | 14–16 years

Representing elements and compounds | Review my learning worksheets | 14–16 years
- Contributors
- Email alerts
Site powered by Webvision Cloud

- school Campus Bookshelves
- menu_book Bookshelves
- perm_media Learning Objects
- login Login
- how_to_reg Request Instructor Account
- hub Instructor Commons
Margin Size
- Download Page (PDF)
- Download Full Book (PDF)
- Periodic Table
- Physics Constants
- Scientific Calculator
- Reference & Cite
- Tools expand_more
- Readability
selected template will load here
This action is not available.

12: Unsaturated and Aromatic Hydrocarbons
- Last updated
- Save as PDF
- Page ID 15304
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
Our modern society is based to a large degree on the chemicals we discuss in this chapter. Most are made from petroleum. Alkanes—saturated hydrocarbons—have relatively few important chemical properties other than that they undergo combustion and react with halogens. Unsaturated hydrocarbons—hydrocarbons with double or triple bonds—on the other hand, are quite reactive. In fact, they serve as building blocks for many familiar plastics—polyethylene, vinyl plastics, acrylics—and other important synthetic materials (e.g., alcohols, antifreeze, and detergents). Aromatic hydrocarbons have formulas that can be drawn as cyclic alkenes, making them appear unsaturated, but their structure and properties are generally quite different, so they are not considered to be alkenes. Aromatic compounds serve as the basis for many drugs, antiseptics, explosives, solvents, and plastics (e.g., polyesters and polystyrene). The two simplest unsaturated compounds—ethylene (ethene) and acetylene (ethyne)—were once used as anesthetics and were introduced to the medical field in 1924. However, it was discovered that acetylene forms explosive mixtures with air, so its medical use was abandoned in 1925. Ethylene was thought to be safer, but it too was implicated in numerous lethal fires and explosions during anesthesia. Even so, it remained an important anesthetic into the 1960s, when it was replaced by nonflammable anesthetics such as halothane (\(\mathrm{CHBrClCF_3}\)).
- 12.1: Prelude to Unsaturated and Aromatic Hydrocarbons The two simplest unsaturated compounds—ethylene (ethene) and acetylene (ethyne)—were once used as anesthetics and were introduced to the medical field in 1924. However, it was discovered that acetylene forms explosive mixtures with air, so its medical use was abandoned in 1925. Ethylene was thought to be safer, but it too was implicated in numerous lethal fires and explosions during anesthesia. Even so, it remained an important anesthetic into the 1960s, when it was replaced by nonflammable anes
- 12.2: Alkenes- Structures and Names Alkenes are hydrocarbons with a carbon-to-carbon double bond.
- 12.3: Cis-Trans Isomers (Geometric Isomers) Cis-trans (geometric) isomerism exists when there is restricted rotation in a molecule and there are two nonidentical groups on each doubly bonded carbon atom.
- 12.4: Physical Properties of Alkenes The physical properties of alkenes are much like those of the alkanes: their boiling points increase with increasing molar mass, and they are insoluble in water.
- 12.5: Chemical Properties of Alkenes Alkenes undergo addition reactions, adding such substances as hydrogen, bromine, and water across the carbon-to-carbon double bond.
- 12.6: Polymers Molecules having carbon-to-carbon double bonds can undergo addition polymerization.
- 12.7: Alkynes Alkynes are similar to alkenes in both physical and chemical properties. For example, alkynes undergo many of the typical addition reactions of alkenes. The International Union of Pure and Applied Chemistry (IUPAC) names for alkynes parallel those of alkenes, except that the family ending is -yne rather than -ene. The IUPAC name for acetylene is ethyne. The names of other alkynes are illustrated in the following exercises.
- 12.8: Aromatic Compounds- Benzene Aromatic hydrocarbons appear to be unsaturated, but they have a special type of bonding and do not undergo addition reactions.
- 12.9: Structure and Nomenclature of Aromatic Compounds Aromatic compounds contain a benzene ring or have certain benzene-like properties; for our purposes, you can recognize aromatic compounds by the presence of one or more benzene rings in their structure.
- 12.10: Unsaturated and Aromatic Hydrocarbons (Exercises) Select problems and solutions for the chapter.
- 12.11: Unsaturated and Aromatic Hydrocarbons (Summary) A brief summary of the chapter.

- school Campus Bookshelves
- menu_book Bookshelves
- perm_media Learning Objects
- login Login
- how_to_reg Request Instructor Account
- hub Instructor Commons
Margin Size
- Download Page (PDF)
- Download Full Book (PDF)
- Periodic Table
- Physics Constants
- Scientific Calculator
- Reference & Cite
- Tools expand_more
- Readability
selected template will load here
This action is not available.

8.13: Physical Properties of Hydrocarbons
- Last updated
- Save as PDF
- Page ID 17356
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
Learning Objectives
- To identify the physical properties of alkanes and describe trends in these properties.
Because alkanes have relatively predictable physical properties and undergo relatively few chemical reactions other than combustion, they serve as a basis of comparison for the properties of many other organic compound families. Let’s consider their physical properties first.
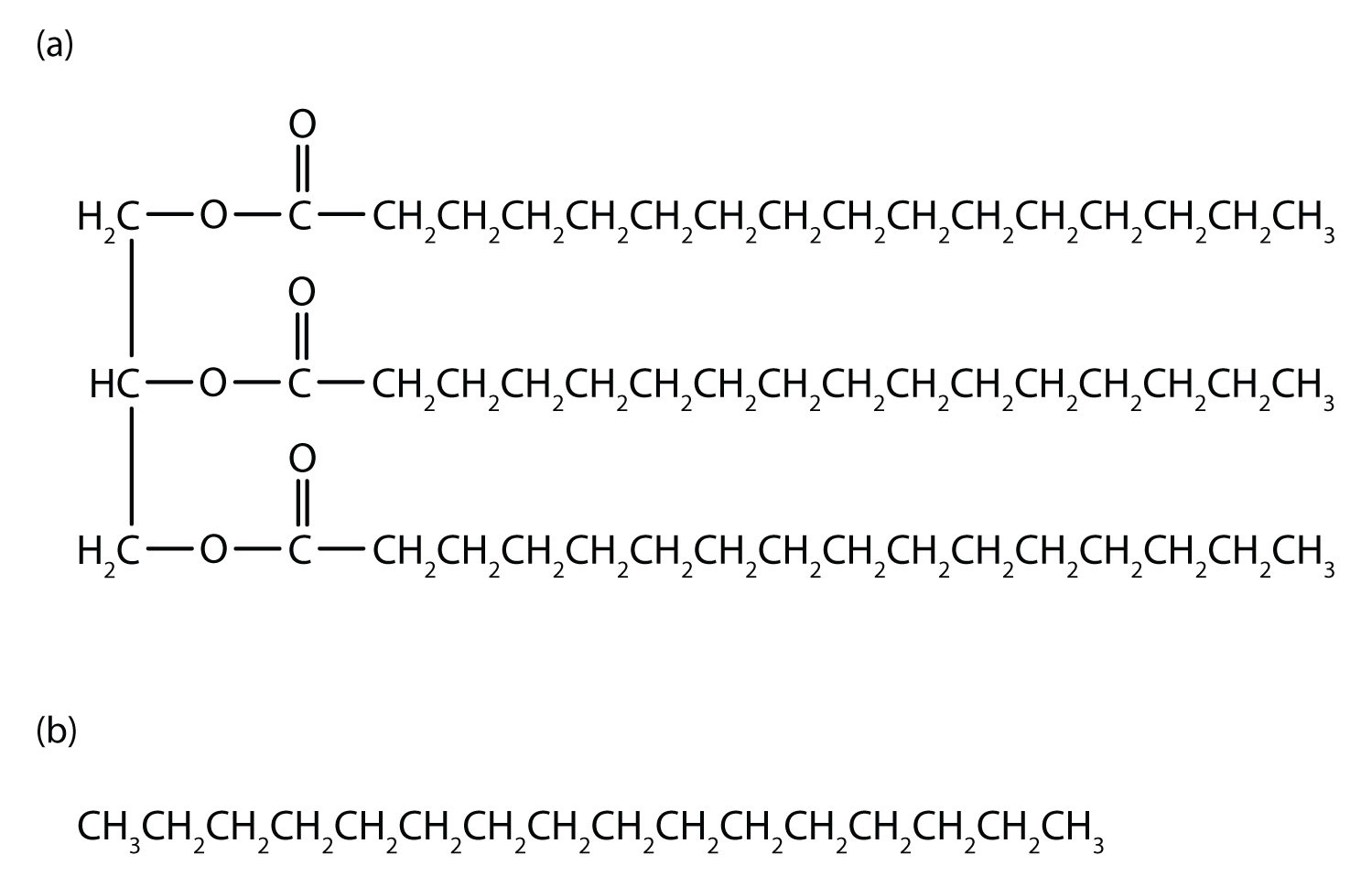
Table \(\PageIndex{1}\) describes some of the properties of some of the first 10 straight-chain alkanes. Because alkane molecules are nonpolar, they are insoluble in water, which is a polar solvent, but are soluble in nonpolar and slightly polar solvents. Consequently, alkanes themselves are commonly used as solvents for organic substances of low polarity, such as fats, oils, and waxes. Nearly all alkanes have densities less than 1.0 g/mL and are therefore less dense than water (the density of H 2 O is 1.00 g/mL at 20°C). These properties explain why oil and grease do not mix with water but rather float on its surface.

Looking Closer: Gas Densities and Fire Hazards
Table \(\PageIndex{1}\) indicates that the first four members of the alkane series are gases at ordinary temperatures. Natural gas is composed chiefly of methane, which has a density of about 0.67 g/L. The density of air is about 1.29 g/L. Because natural gas is less dense than air, it rises. When a natural-gas leak is detected and shut off in a room, the gas can be removed by opening an upper window. On the other hand, bottled gas can be either propane (density 1.88 g/L) or butanes (a mixture of butane and isobutane; density about 2.5 g/L). Both are much heavier than air (density 1.2 g/L). If bottled gas escapes into a building, it collects near the floor. This presents a much more serious fire hazard than a natural-gas leak because it is more difficult to rid the room of the heavier gas.
Also shown in Table \(\PageIndex{1}\) are the boiling points of the straight-chain alkanes increase with increasing molar mass. This general rule holds true for the straight-chain homologs of all organic compound families. Larger molecules have greater surface areas and consequently interact more strongly; more energy is therefore required to separate them. For a given molar mass, the boiling points of alkanes are relatively low because these nonpolar molecules have only weak dispersion forces to hold them together in the liquid state.
Looking Closer: An Alkane Basis for Properties of Other Compounds
An understanding of the physical properties of the alkanes is important in that petroleum and natural gas and the many products derived from them—gasoline, bottled gas, solvents, plastics, and more—are composed primarily of alkanes. This understanding is also vital because it is the basis for describing the properties of other organic and biological compound families. For example, large portions of the structures of lipids consist of nonpolar alkyl groups. Lipids include the dietary fats and fatlike compounds called phospholipids and sphingolipids that serve as structural components of living tissues. These compounds have both polar and nonpolar groups, enabling them to bridge the gap between water-soluble and water-insoluble phases. This characteristic is essential for the selective permeability of cell membranes.
Key Takeaway
- Alkanes are nonpolar compounds that are low boiling and insoluble in water.

IMAGES
VIDEO
COMMENTS
The three types of unsaturated hydrocarbons is alkynes, alkenes, and aromatic hydrocarbons. Their boiling points tend to increase with chain length. The boiling point of 1-octyne is higher than 1-pentyne. The structure has 7 carbons. 3 is the first carbon in the triple bond.
Study with Quizlet and memorize flashcards containing terms like On a piece of paper, draw the structure of 3-heptyne. Now describe the structure you have drawn:, On a piece of paper, draw the structure of cis-4-nonene. Now describe the structure you have drawn:, Use the drop-down menus to name the following structures. and more.
Compounds containing double or triple bonds are often referred to collectively as unsaturated compounds. Because of their multiple bonds, alkenes and alkynes are usually more chemically reactive than alkanes and aromatic hydrocarbons. The presence of a double or triple bond in the molecule opens up many more possibilities for isomerism than is ...
Unsaturated hydrocarbons have double or triple bonds and are quite reactive; saturated hydrocarbons have only single bonds and are rather unreactive. ... How do these properties compare to those of the alkanes? Without consulting tables, arrange the following alkenes in order of increasing boiling point: 1-butene, ethene, 1-hexene, and propene. ...
As noted before, alkenes are hydrocarbons with carbon-to-carbon double bonds (R 2 C=CR 2) and alkynes are hydrocarbons with carbon-to-carbon triple bonds (R-C≡C-R).Collectively, they are called unsaturated hydrocarbons because they have fewer hydrogen atoms than does an alkane with the same number of carbon atoms, as is indicated in the following general formulas:
Hydrocarbons are compounds that contain only carbon and hydrogen. The carbon atoms in hydrocarbons may share single, double, or triple covalent bonds. Unsaturated hydrocarbons contain at least one double or triple bond between carbon atoms. They are classified on the basis of their bonds as alkenes, aromatic hydrocarbons, or alkynes.
These are complex organic molecules with long chains of carbon atoms, which contain at least one double bond between carbon atoms. Unsaturated hydrocarbon molecules that contain one or more double bonds are called alkenes. Carbon atoms linked by a double bond are bound together by two bonds, one σ bond and one π bond.
This chain is the parent alkene. It has the root. name of the alkane with the same number of carbons plus the ending -ene. The chain is numbered so that the carbon atoms of the double bond have the lowest possible numbers. Substituents on the chain are named and num-bered in the same way they are for alkanes.
A. Use the drop-down menus to classify each of the following structures into one of the four types of hydrocarbons. 1. alkyne. 2. alkane. 3. aromatic hydrocarbon. 4. alkene. Study the graphs. Identify the graph that best represents the trend in alkene boiling point as a function of carbon chain length. graph A.
In Chapter 12 "Organic Chemistry: Alkanes and Halogenated Hydrocarbons" we noted that alkanes—saturated hydrocarbons—have relatively few important chemical properties other than that they undergo combustion and react with halogens. Unsaturated hydrocarbons—hydrocarbons with double or triple bonds—on the other hand, are quite reactive.
Unsaturated hydrocarbons are extremely useful organic compounds in the manufacturing of plastics. Polystyrene is used in making egg cartons, disposable cups, and other convenient products. Industrial chemicals such as alcohol include the usage of alkenes in their manufacturing process. Some unsaturated hydrocarbons are used as general anaesthetics.
Double and triple bonds give rise to a different geometry around the carbon atom that participates in them, leading to important differences in molecular shape and properties. The differing geometries are responsible for the different properties of unsaturated versus saturated fats. Ethene, C 2 H 4, is the simplest alkene. Each carbon atom in ...
Exercises. Write the condensed structural formula of the monomer from which Saran is formed. A segment of the Saran molecule has the following structure: CH 2 CCl 2 CH 2 CCl 2 CH 2 CCl 2 CH 2 CCl 2. Write the condensed structural formula for the section of a molecule formed from four units of the monomer CH 2 =CHF.
4.7.1.3 Properties of hydrocarbons. Some properties of hydrocarbons depend on the size of their molecules, including boiling point, viscosity and flammability. These properties influence how hydrocarbons are used as fuels. Students should be able to recall how boiling point, viscosity and flammability change with increasing molecular size.
11.1: Prelude to Unsaturated and Aromatic Hydrocarbons. The two simplest unsaturated compounds—ethylene (ethene) and acetylene (ethyne)—were once used as anesthetics and were introduced to the medical field in 1924. However, it was discovered that acetylene forms explosive mixtures with air, so its medical use was abandoned in 1925.
12.1: Prelude to Unsaturated and Aromatic Hydrocarbons. The two simplest unsaturated compounds—ethylene (ethene) and acetylene (ethyne)—were once used as anesthetics and were introduced to the medical field in 1924. However, it was discovered that acetylene forms explosive mixtures with air, so its medical use was abandoned in 1925.
distinguish between saturated and unsaturated hydrocarbons. We will investigate some of the chemical and physical properties of hydrocarbons and their. derivatives and use their properties to distinguish between: alkanes, alkenes, and aromatics. Below, is a comparison of physical appearance of two structural isomers of dichlorobenzenes
an unsaturated hydrocarbon. 2. Briefly identify the important distinctions between an alkene and an alkane. 3. Classify each compound as saturated or unsaturated. Identify each as an alkane, an alkene, or an alkyne. 1. 2. CH. 3. CH. 2. C≡CCH. 3. 3. Answers. 1. Unsaturated hydrocarbons have double or triple bonds and are quite reactive;
The 1 is not necessary. The name of the compound is simply propene. 12.1: Hydrocarbons. The simplest organic compounds are hydrocarbons and are composed of carbon and hydrogen. Hydrocarbons can be aliphatic or aromatic; aliphatic hydrocarbons are divided into alkanes, alkenes, and ….
Summary Properties and Uses of Saturated Hydrocarbons Alkanes and Their Uses • Shapes of : straight chain, branched chain, and cycloalkanes • Properties: solubility, point, and reactivity • Uses of hydrocarbons: gasoline, natural gas, oil, and asphalt Use this space to write any questions or thoughts about this lesson. 2 Slide
Table 8.13.1 8.13. 1 describes some of the properties of some of the first 10 straight-chain alkanes. Because alkane molecules are nonpolar, they are insoluble in water, which is a polar solvent, but are soluble in nonpolar and slightly polar solvents. Consequently, alkanes themselves are commonly used as solvents for organic substances of low ...